svs vQI reporting
The SVS PSO (Patient Safety Organization) produces several SVS VQI reports that are designed to facilitate and drive quality improvement efforts for SVS VQI centers and regional quality groups.
Presentations are provided below as supplementary material to aid centers in understanding and interpreting the reports. Users are encouraged to view these presentations in “Slide Show” mode for maximum benefit, where narrative text is provided in the “Notes” section of each slide to further explain and clarify content.
- An Introduction to PSO Reporting – A high-level introduction and overview of the various reports
- Drilling Into Quarterly Dashboards – A focused presentation on the quarterly VQI Best Practices Dashboard report

svs vqi reporting schedule
The SVS VQI Reporting Schedule for 2023-2024 is now available. Please remember to check the reporting schedule for important data-entry/completion deadlines associated with each SVS VQI report.
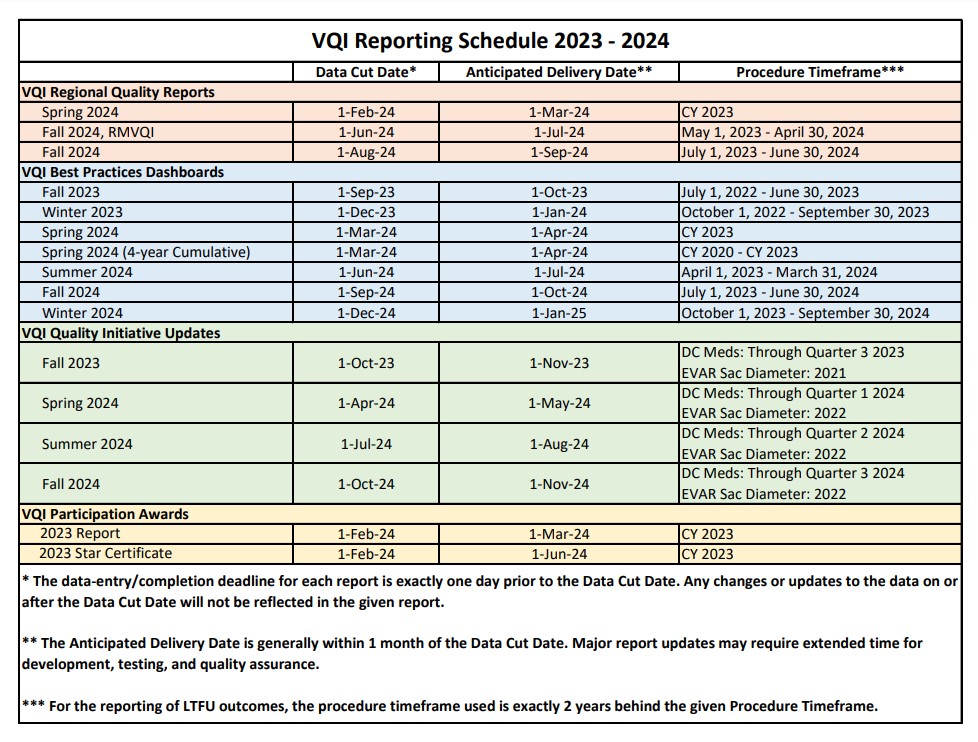
VQI REGIONAL QUALITY REPORTS
The regional quality reports are the cornerstone of the SVS VQI’s spring and fall regional quality group meetings. These reports show centers where they stand in comparison to other centers in their region and to all centers in the SVS VQI on these 32 key measures:
- Multiple registries: Long-Term Follow-up
- Multiple registries: Discharge Medications
- Multiple registries: Preop Smoking
- Multiple registries: Smoking Cessation at Follow-Up
- TFEM CAS ASYMP: Stroke/Death
- TFEM CAS SYMP: Stroke/Death
- TCAR ASYMP: Stroke/Death
- TCAR SYMP: Stroke/Death
- CEA ASYMP: Stroke/Death
- CEA ASYMP: Postop LOS>1 Day
- CEA SYMP: Stroke/Death
- CEA SYMP: Postop LOS>1 Day
- EVAR: Postop LOS>2 Days
- EVAR: Sac Diameter Reporting
- EVAR: SVS Sac Size Guideline
- TEVAR: Sac Diameter Reporting
- OAAA: In-Hospital Mortality
- OAAA: SVS Cell-Saver Guideline
- PVI CLAUD: ABI/Toe Pressure
- INFRA: ABI/Toe Pressure
- INFRA CLAUD: Return to OR
- INFRA CLTI: Return to OR
- INFRA CLTI: WIfI Documentation
- SUPRA: ABI/Toe Pressure
- SUPRA CLAUD: Return to OR
- SUPRA CLTI: Return to OR
- SUPRA CLTI: WIfI Documentation
- LEAMP: Postop Complications
- HDA: Primary AVF vs. Graft
- HDA: Ultrasound Vein Mapping
- HDA: Postop Complications
- IVCF: Filter Retrieval Reporting
Each of these reports provides information on the center’s performance during the previous year and over the past 4 years, comparing the center’s rates with the rates of other centers in the region and all centers in the SVS VQI.
VQI Best Practices Dashboards
The Best Practices Dashboards provide each center with its rate on more than 200 outcomes (including results for the DC Medications national quality initiative) and process measures across 14 SVS VQI registries, comparing that rate with regional and national benchmarks. Color-coding is provided to make it easy for center staff to whether they are in the top 25% of all centers (blue) or the bottom 25% (coral). The outcomes reported in the dashboards are chosen by the governing committees for each registry and include results for the DC Medications national quality initiative.
VQI Quality Initiative Updates
The SVS PSO, in consultation with the Arterial Quality Council, selects SVS VQI-wide quality improvement initiatives. The goals of these initiatives are publicized to all member centers and discussed at spring and fall regional quality meetings. Current measures are prescription of antiplatelet and statin at discharge after all vascular procedures, and reporting of sac diameters between 9-21 months after EVAR. Performance updates on the quality initiatives are delivered quarterly to each center, giving each center its progress to date on these initiatives along with national benchmarks. The QI updates provide rapid data-feedback for centers to evaluate their data in preparation for the yearly participation awards, where centers can earn additional credit for exemplary participation in national quality improvement initiatives.
